
Calculate the pH and the equilibrium concentrations of H2C6H5O7-, HC6H5O72-, and C6H5O73- in a 0.1380 M aqueous citric acid solution. For H3C6H5O7, Ka1 = 7.4 x 10-3, Ka2 = 1.7 x 10-5, and Ka3 = 4.0 x 10-7. | Homework.Study.com

Clean Green 1 Gal. Citric Acidifier - Concentrated Liquid Citric Acid Solution - pH Down for Cleaning, Agriculture and More CA-128 - The Home Depot

SOLVED:Citric acid, which is present in citrus fruits, is a triprotic acid (Table 16.3). (a) Calculate the pH of a 0.040 M solution of citric acid. (b) Did you have to make

SOLVED: Ve5 NN-N V €= 5 #6 6 # € = VG # €= Outali Chovge What is the pH of a solution containing 0.2 M citric acid (pK, = 5.4) and
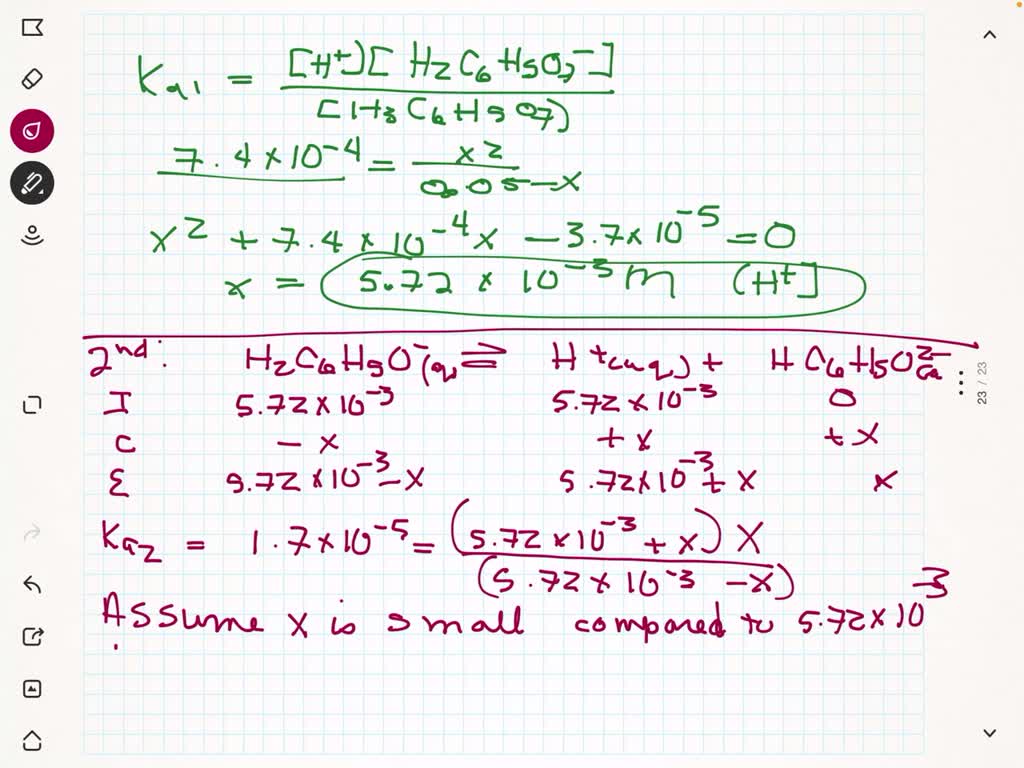
SOLVED:Citric acid, which is present in citrus fruits, is a triprotic acid (Table 16.3). Calculate the pH and the citrate ion (C6 H5 O7 ^3) concentration for a 0.050 M solution of

Calculate the pH and the equilibrium concentrations of H2C6H5O7-, HC6H5O72-, and C6H5O73- in a 0.1380 M aqueous citric acid solution. For H3C6H5O7, Ka1 = 7.4 x 10-3, Ka2 = 1.7 x 10-5, and Ka3 = 4.0 x 10-7. | Homework.Study.com

Lemon juice normally has a `pH` of `2`. If all the acid the lemon juice is citric acid and there are - YouTube
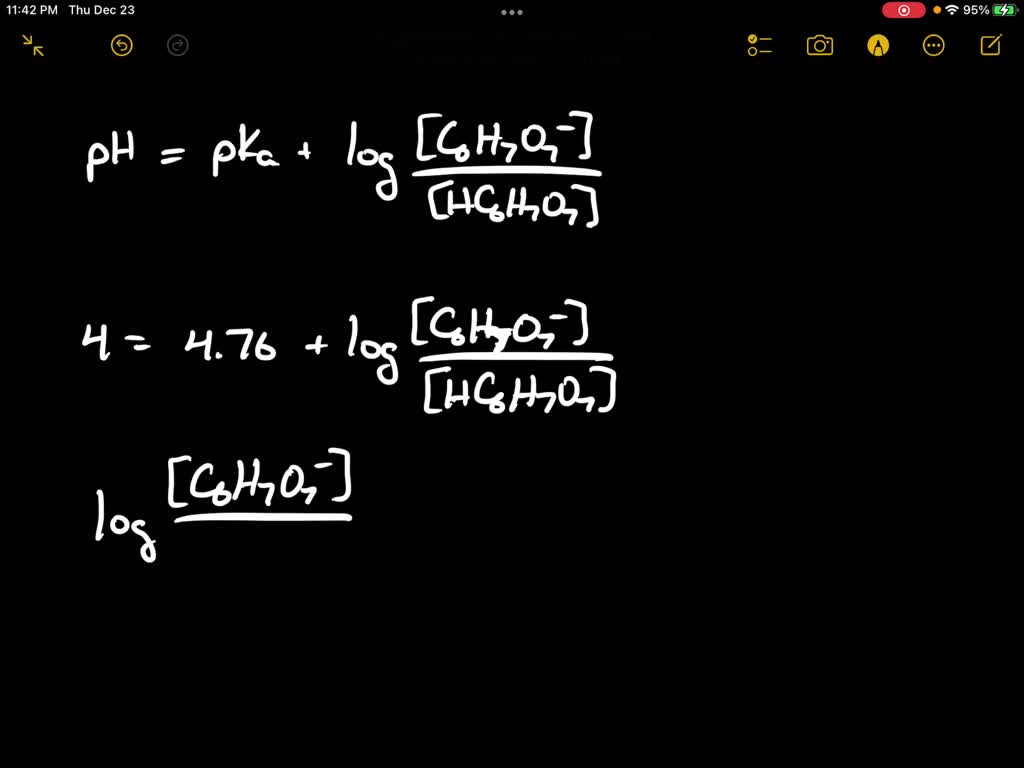
SOLVED: The pH of the 0.5% w/v citric acid solution is 4. Citric acid is a weak acid, pka=4.76. (Mol wt: 192g/mole) a. Calculate the % ionization of diluted citric acid solution

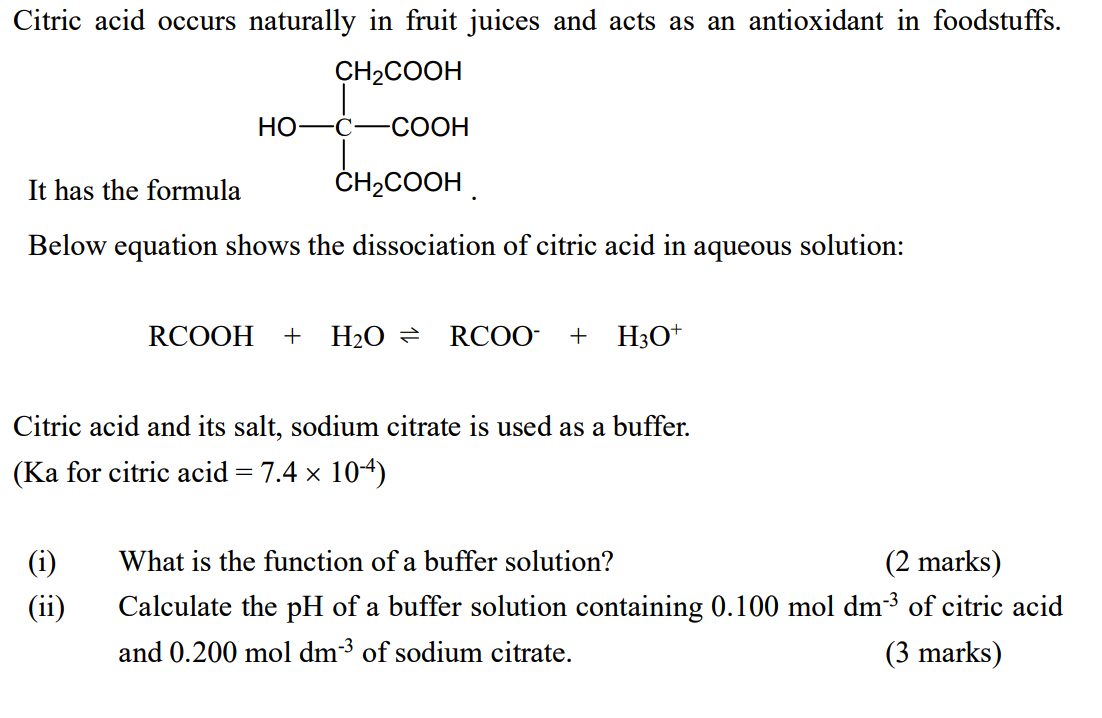


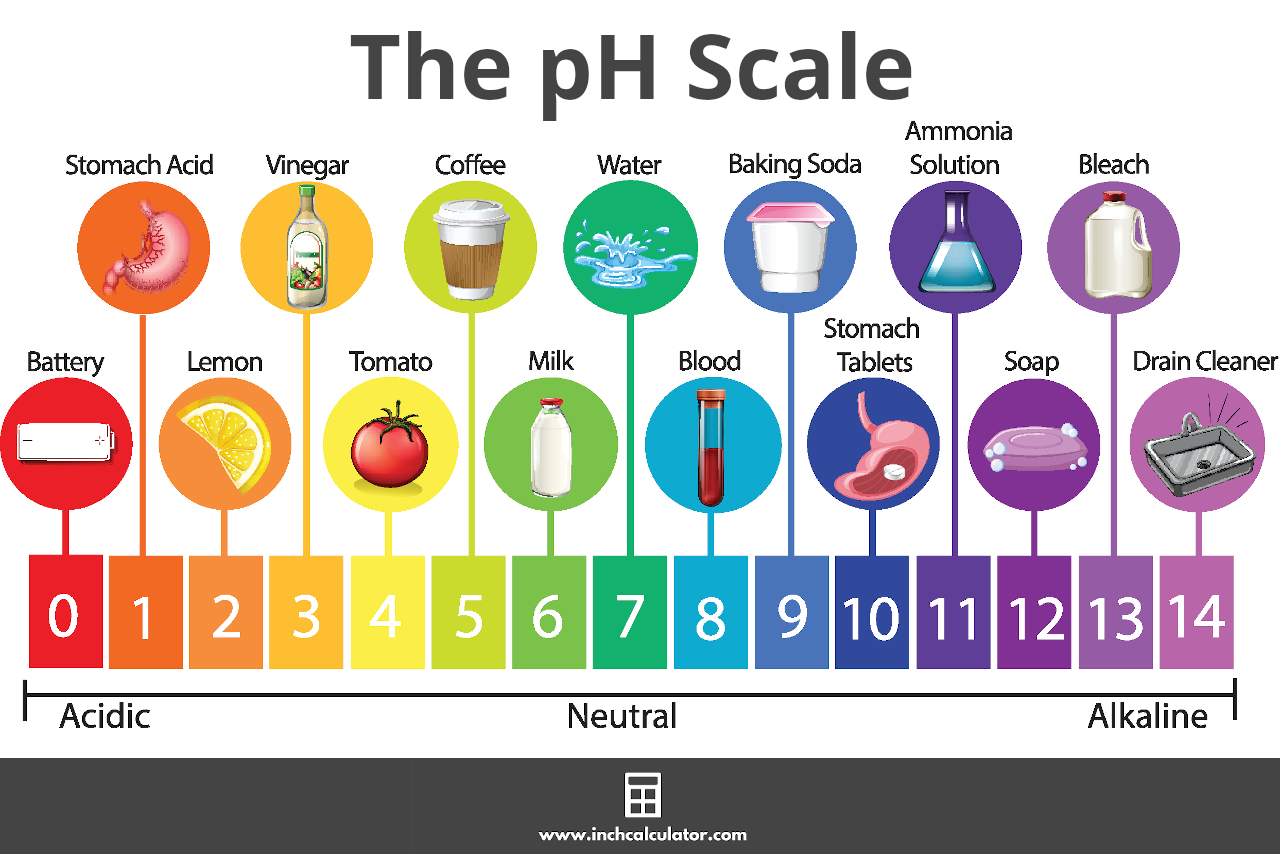






![Solved 6 SOT 4. a) [2] One litre of buffer is made by mixing | Chegg.com Solved 6 SOT 4. a) [2] One litre of buffer is made by mixing | Chegg.com](https://media.cheggcdn.com/study/e68/e687f593-aceb-416b-bd35-b4e070da579b/image.png)

